In the last year, I’ve tried more than 50 hearing aids—from brands I’d never heard of on Amazon to well-known names like Sennheiser and Jabra. I used their smartphone apps, took notes on comfort and ease of use, and worked with Soundly’s audiology team to test them with professional audiological equipment.
What did I find? Some OTC hearing aids can go toe-to-toe with prescription brands you’d find in clinics for 2–3x the price. Others? They’re not much help at all.
In this guide, we’ll break it all down: our favorite products, the pros and cons, and what you need to know to decide between OTC and prescription options.
Let’s dig in.
Prefer to watch? Here's my recently updated video guide to the OTC hearing aid market in 2026. 👇
OTC Hearing Aids are Shifting the Industry
Formalized by the FDA in 2022, they offer quality technology at more affordable prices, making hearing care more accessible than ever.
Made by Leading Global Brands
Top OTC hearing aids are produced by manufacturers like WSA and Sonova, the same companies behind many in-clinic hearing aids.
Ideal for Mild to Moderate Hearing Loss
OTC devices are designed for specific levels of hearing loss. Take our hearing test to find out where you stand.
Some Tech Savvy Required
Most OTC hearing aids need basic smartphone skills for setup and programming through their companion apps.
- 50+ hearing aid brands reviewed and rated by our team of hearing aid wearers and audiologists
- 200+ hours each month spent researching brands and care options
- 2,000,000 people shopped on Soundly in 2024
- 100% independently owned and operated
Read more about our company, services and process here.
Featured in this article
Our Top OTC Hearing Aid Picks for 2026
After testing dozens of options, we’ve narrowed our list to four standout over-the-counter products that offer exceptional value and performance. Each device is made by a trusted manufacturer, delivers strong sound quality, and includes Soundly’s ongoing care. Importantly, the list covers a range of prices and styles to suit different needs.
- Sennheiser All Day Clear – Behind-the-ear comfort, Bluetooth streaming, and standout sound quality from a top-tier manufacturer.
- Eargo 8 – Nearly invisible, in-ear design with great sound and all-day comfort—minus Bluetooth.
- Nuance Audio Glasses – Stylish glasses with built-in speakers, ideal for mild hearing loss and open-ear comfort.
- ELEHEAR Beyond – Feature-packed budget option with Bluetooth, rechargeability, and solid performance under $400.
Sennheiser All Day Clear
- Our favorite OTC hearing aids for under $1,000
- Manufactured by the largest hearing aid maker in the world
- Compatible with iPhone and Android devices
Sennheiser All Day Clear is our top-selling and best-rated OTC hearing aid at Soundly.com. We’ve tested this product extensively—in the audiology test box, in real-world noisy environments, and through thousands of Soundly customer experiences in 2025.
Soundly offers a complimentary setup call with every purchase — a helpful perk for customers and a valuable way for us to hear firsthand which products are performing best.
At-Home Delivery

Sennheiser All Day Clear is made by Sonova, the world’s leading hearing aid manufacturer. Unlike traditional in-clinic devices that average around $5,000 a pair, All Day Clear ships directly to your home (Soundly offers free 2-day shipping) and lists for $999 per pair.
Rechargeable with Bluetooth
These hearing aids deliver 18–20 hours of battery life on a single charge and offer reliable call and music streaming for both iPhone and Android users. Thanks to Sonova’s Bluetooth Classic connection, pairing is easier than most OTC devices—especially for Android users.
App Programming
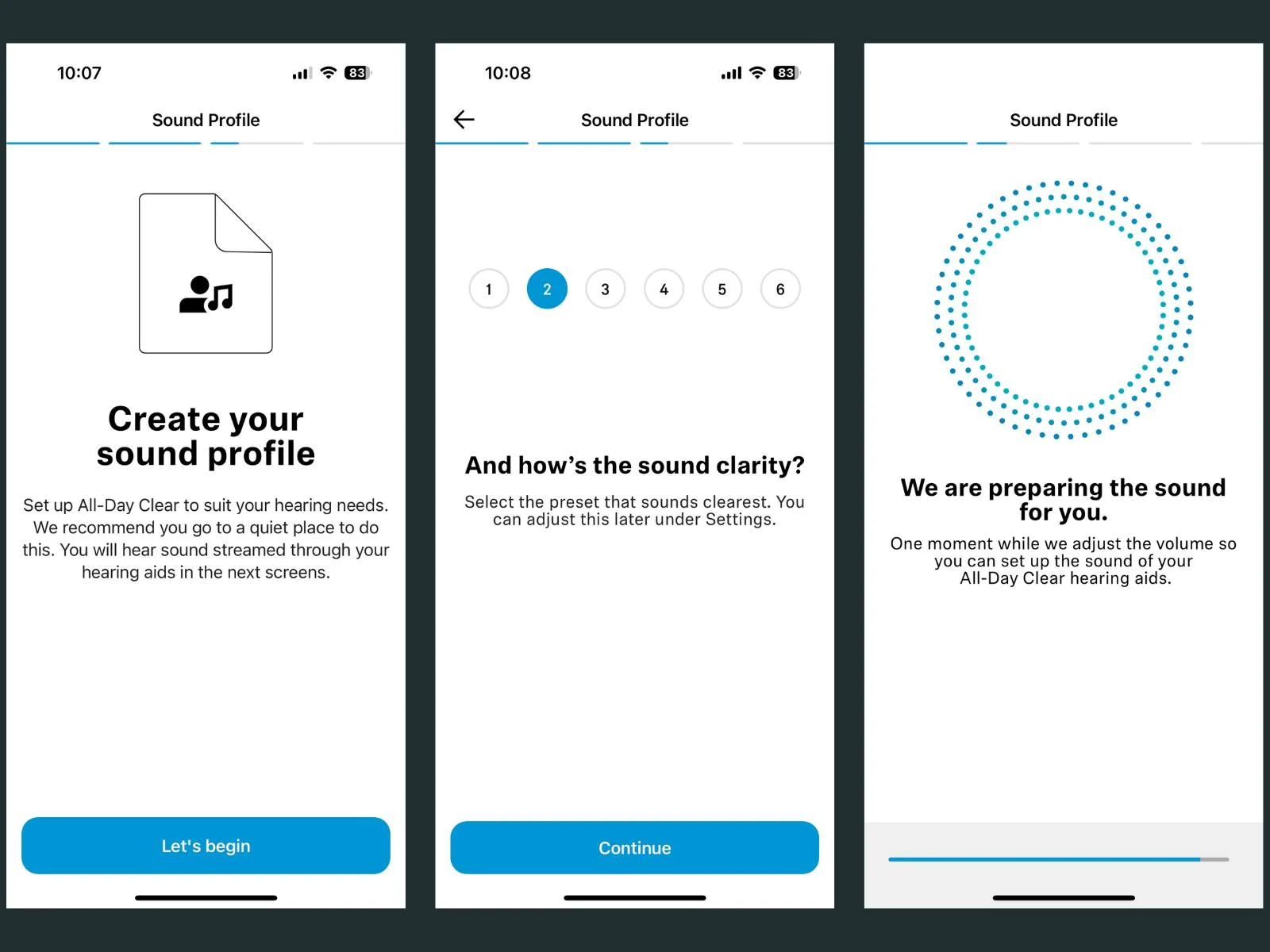
As a self-fitting product, All Day Clear uses a smartphone app to adjust the hearing aids to your specific hearing loss. The app isn’t the most feature-rich on the market, but in our testing (and from what we hear from customers), it’s the easiest to set up and navigate.
Sound Quality
Likely your biggest question: how do they sound? In short—excellent. You can hear Sonova’s signature processing in these devices. They handle sudden sounds, wind, feedback, and background noise in a way we’d consider market-leading for OTC self-fitting hearing aids.
Notes & Watchouts
- Best suited for mild-to-moderate hearing loss. If your loss is more significant or complex, in-clinic care is still the right move. Take our hearing test to better understand your loss.
- The charger must be plugged into the wall while in use. A separate power pack is available if you want mobile charging.
Bottom line: Sennheiser All Day Clear sits at the top of our list for self-fitting OTC hearing aids. It delivers great sound, simple control, and strong value from a world-class manufacturer.
You can purchase Sennheiser All Day Clear hearing aids directly through Soundly for $999 per pair and access all of our care and services.
We recommend taking our hearing test before purchasing. Our team can review your results and offer a second opinion if needed.
Once you’re ready, you can place your order on the website or over the phone.
- Sennheiser orders come with 100-day risk-free returns — exclusively on Soundly.
- Your hearing aids ship 2nd Day Air to your home.
- Upon arrival, you’ll receive setup instructions and use the smartphone app to customize your hearing aids.
- Sennheiser includes starter domes in the box; you can purchase alternate sizes and styles through Soundly.
- Every order includes a complimentary setup session with one of our hearing professionals.
- We offer MSRP pricing and deals and will price-match any other official retailer offering the same product and terms.
- Soundly provides 45-day price-drop protection—if the price drops within 45 days, contact us and we’ll refund the difference.
Here's what Dr. Amy Sarow, Audiology Lead at Soundly, had to say about the Sennheiser All Day Clear:
What is your experience supporting customers directly with this product. What comes up a lot?
Customers often ask about how to adjust their devices to match or closely align with their audiogram. While OTC devices use ‘presets’ - if they have a mild to moderate high frequency loss, we can usually come close to what is appropriate for their needs.
The Sound Profile uses three categories during the setup: volume, clarity, and balance. We generally perceive volume in the low frequencies, most people have normal hearing to mild hearing loss in that region, so the preset default of 3 is usually fine.
The clarity setting is the amount of high frequency amplification. The default here is also 3, which is appropriate for a mild loss and for a moderate loss a high number is more appropriate (i.e., a 5 or 6). The balance setting allows the user to adjust volume for right and left separately.
Customers often ask how to keep the adjustments when they ‘split’ the volume on the main app screen, but this setting will revert back to the middle after the hearing aids are placed in the charger and taken out again the next day.
Compared with other hearing aids at a similar price, where does Sennheiser All Day Clear stand out, and where does it fall short?
Compared to other products at the same price point, Sennheiser All Day Clear shares the processing of its prescription counterparts (Unitron and Phonak).
The sound processing feels smoother and more sophisticated than some other options in this pricepoint or lower.
The ADC also does an excellent job of feedback suppression, with some other similar OTC products more prone to feedback in the ear.
However, the ADC app falls a bit short compared to some of the other OTC apps. For example, no direct controls in the app for background noise suppression, ability to directly select hearing aid programs.
The app feels a bit more simplistic than some comparable OTC brand apps.
Who is this product best for?
This product is best for those who have mild to moderate hearing loss and works particularly well for a sloping hearing loss configuration.
For those with normal hearing in the low frequencies, they may experience some static or white noise sound, but this can be reduced by adjusting the ‘mid’ category down on the equalizer.
Additionally, this product works well for those who like to stream audio via Bluetooth, works better than other products for those who like to listen to music, and is a great option for those who prefer an app that keeps things simple.
It’s not always the best choice for glasses wearers (though still possible) or those with very petite ears.
- Premium Sonova-built technology for less than prescription channels
- Bluetooth streaming and hands-free calls (Android and iPhone)
- Comfortable all-day form factor
- Charger must be plugged in while charging
- Requires a smartphone for programming
- All-Day Clear is an OTC product appropriate for mild-moderate hearing loss levels
- If you have more significant hearing loss you might consider local or Telehealth care. Read more about prescription Vs. OTC hearing aids here
- Available for purchase online via Soundly
- Does not require a prescription from a doctor
- Customize your hearing aids with the Sennheiser All-Day Clear app
- Soundly customers get a complimentary guided setup session
- Sennheiser offers impressive underlying background noise management technology
- We found this product performed at a similar level to top prescription brands
- Comfortable receiver-in-canal style is great for all day wear
- Sits behind the ear with a receiver (speaker) that reaches into the ear canal
- The form factor of Sennheiser All-Day Clear is comparable to leading prescription hearing aids like Phonak Lumity
- Available in a dual-tone black and silver
- Choose between slim and non-slim versions (we recommend the non-slim option for most people)
- Sennheiser All-Day Clear comes with rechargeable batteries
- 16 hours of wear on a single charge
- Recharge case must be plugged into the wall while in use
- All-Day Clear streams to all devices including iPhone and Android
- All Day Clear allows hands free phone calls on both iPhone and Android
- Streaming quality is similar to the top prescription products on the market
- All-Day Clear is reasonably easy to handle but does require the wearer to place the hearing aid behind their ear and insert the receiver into the ear (consistent across all behind-the-ear styles)
- The All-Day Clear recharge case has ports that must be in contact for charging. This type of charger is more difficult than premium conduction chargers which do not require ports to be touching
- Sennheiser All-Day Clear hearing aids have an IP68 rating
- These devices can be submerged in up to one meter of water for 30 minutes without damage
- The Sennheiser All-Day Clear app enables setup and customization of your hearing aids
- The smartphone app allows on-the-go volume and sound quality adjustments
- The All-Day Clear app has a music and automatic program but does not offer Tinnitus masking, program creation or other advanced features
Eargo 8
- Disruptive leader in hearing health, known for sleek, invisible design.
- Rechargeable with a patented ear tip that allows ears to breathe.
- Eargo 8 adapts to your environment automatically throughout the day.
- Budget models (Eargo SE and Eargo LINK) offer lower prices and alternate features.
Eargo has long set the standard for invisible-style design in the hearing aid industry. Their compact devices, along with an intuitive app and thoughtful packaging, make Eargo a standout choice for early adopters. Here’s why we picked it:
- Invisible Design: The Eargo 8 is one of the smallest rechargeable hearing aids on the market, rivaled only by the Sony CRE-C20.
- Reduced Occlusion: Eargo’s patented “floating” ear tip design sits comfortably in the ear without causing occlusion, a common issue for in-ear hearing aid users.
- User-Friendly App: The Eargo app is easy to navigate, and it allows audiologists to remotely adjust your devices as needed, though most adjustments can be managed directly in the app.
Eargo has received numerous customer reviews, reflecting a generally positive reception.
Overall
Customers frequently commend Eargo for its discreet design, sound quality, and responsive customer service. Many users highlight the significant improvement in their hearing experience and the comfort of the devices.
Positives
- Discreet Design: "The Eargo hearing aids are virtually invisible, and no one can tell I'm wearing them." — James D.
- Sound Quality: "The clarity of sound is remarkable; I can hear conversations clearly even in noisy environments." — Charlie B.
- Customer Service: "Eargo's support team was incredibly helpful and guided me through the setup process with ease." — Rowdy G.
Complaints
While the majority of feedback is positive, some customers have reported issues with device durability and the need for adjustments. However, these concerns are relatively infrequent compared to the overall satisfaction expressed by users.
For a comprehensive view of customer experiences, you can visit Eargo's review page.
- Eargo is a tiny, rechargeable device that sits entirely inside the ear
- Eargo uses a unique design that prevents occlusion
- Eargo is self-fit using an app but offers remote support
- Only appropriate for those with mild to moderate hearing loss
- Requires some tinkering to fine tune the product
- Eargo is not Bluetooth enabled due to the tiny size
- Eargo 8 hearing aids are sold over the counter and are appropriate for those with mild-moderate hearing loss
- If you have more significant hearing loss it is recommended that you access professional care through Telehealth or local care
- Available for purchase online or at retail stores like Verizon Victra and Best Buy
- Does not require a prescription from a doctor
- Customize your hearing aids with an onboard hearing test and app-controls
- Eargo has well-developed background noise management algorithms
- Smart Sound Adjust automatically adapts to your environment and focuses on voices
- Eargo sits inside of the ear and is one of the most comfortable in-ear options on the market
- Eargo creates less occlusion than many in-the-ear options
- Our team finds all in-the-ear models slightly less comfortable than RIC hearing aids
- Sit inside the ear
- Only available in one completely-in-canal size (nearly invisible in many ears)
- Only available in black
- Eargo hearing aids are rechargeable
- 16 hours of battery life on a single charge
- Each pair of hearing aids comes with a small and portable recharge case that carries 14 additional charges
- Eargo 8 does not offer Bluetooth streaming
- Eargo 8 is small but comes with rechargeable batteries which prevents difficult battery changes
- Eargo's devices include a pull-tab that makes it easier to place and remove the devices in your ears
- Eargo 8 hearing aids have an IP68 rating, meaning they can be submerged in up to one meter of water for 30 minutes without damage.
- Eargo 8 pairs with a smartphone app that includes an onboard hearing test and manual controls for programs, volume, and sound quality.
- You can also adjust volume and switch programs without the app by simply double-tapping your ear.
Nuance Audio Glasses
- Glasses and hearing aids combined
- From the maker of Ray-Ban
- Easy-to-use smartphone app
- Available with multiple style, lenses and colors
Nuance Audio Glasses are one of the most exciting new entries into the hearing aid space.
- Hearing Aids + Glasses: The product combines market-leading eyewear design with a fully functional, open-ear hearing aid.
- Directional Mics: Onboard microphones detect the direction you're facing and deliver real-time amplification of voices directly into your ears.
- Open Ears: Importantly, the glasses don’t go in or over your ear canals, offering both a discreet appearance and a more comfortable, open-ear listening experience.
Nuance is a great option for those with mild-moderate hearing loss or anyone who prefers not to wear traditional hearing aids. The product is available through Soundly here and comes with our full suite of care and support.
Customer feedback on Nuance Glasses has been largely positive, with many users highlighting the open-ear speaker design as comfortable and easy to get used to.
Several people note that the glasses provide clear audio in situations where they would otherwise struggle, making everyday conversations and activities more accessible.
Soundly customers also appreciate the discreet, invisible nature of the device, along with the added convenience of pairing the glasses with prescription or transition lenses.
- Sleek, glasses-style form factor — no in-ear components
- Transitions® lenses included (gray or emerald tint depending on frame color)
- Compatible with prescription lenses via your local optometrist
- Built-in speakers and microphones for discreet hearing support
- No music or media streaming capability
- Battery lasts 8–10 hours — may require midday charging
- Not as powerful as traditional in-ear hearing aids
- Nuance Audio hearing glasses are sold over the counter and are appropriate for those with mild-moderate hearing loss
- If you have more significant hearing loss it is recommended that you access professional care through Telehealth or local care
- Available for purchase here Soundly.com with a complimentary guided setup service
- Does not require a prescription from a doctor
- Customize your hearing glasses using a smartphone app
- Swap your transition lenses for prescription lenses at a local optometrist (prescription lenses not included with purchase)
- Nuance Audio Glasses perform well in background noise (especially in front of you)
- Nuance Audio Glasses leave your ear canals open which can help promote natural awareness
- Nuance Audio Glasses are very comfortable for all day wear due to their open ear design
- Amplification comes through an open speaker placed above the ear canal
- Nuance Audio is the only FDA-cleared pair of hearing glasses on the market at this time
- Nuance Audio Glasses are charged using a charge pad and USBC charger
- Each charge give the user 8-10 hours of wear time (depending on background noise)
- Nuance Audio Glasses do not offer Bluetooth streaming
- Nuance Audio Glasses are among the easiest hearing devices to manage
- No placement of small pieces inside the ear necessary
- Easy charge-pad with bright light to indicate charging
- Protected from sweat, light rain and dust
- Nuance Audio glasses pair with a smartphone app for easy volume control, program updates, and initial calibration
Elehear Beyond
- Our favorite OTC hearing aids for under $500
- Compatible with iPhone and Android devices
- Bluetooth streaming and rechargeable
Here’s why we picked Elehear Beyond:
- Sound Clarity: Elehear Beyond’s noise reduction effectively separates speech from background sounds, providing clear listening in a variety of environments.
- Simple Bluetooth Streaming: Elehear Beyond connects directly through Bluetooth settings, similar to pairing regular headphones. It supports music and calls without requiring extra accessibility setup steps.
- Battery Life: The charging case provides up to 20 hours of use after a 90-minute charge, making it convenient for all-day wear without needing frequent recharges.
- Customizable Sound Settings: Through the Elehear app, users can adjust bass, treble, and volume to match their personal preferences, offering more control than pre-set options in higher-end models.
- Affordable Choice: Priced at $399 per pair, Elehear Beyond is an accessible option in the OTC market, with essential features suited for those who prefer handling adjustments independently.
- "These are amazing for the price! I bought them as a backup to my expensive prescription ones and I use them more often than the originals. The price is amazing for what you get. One suggestion is that there should be sport guards for them to protect from loss"
- "Good hearing aids for the price. They’re effective, but I’ve had some minor issues with background noise in crowded areas. Nothing major, but something to note"
- "I've had the Beyond hearing aids for a few weeks now. They work well, but I’m still getting used to adjusting the settings. Overall, a solid product, but a bit of a learning curve."
- "Thanks to customer support.My hearing aids arrived damaged, customer support was able to replace it in just one day"
- "Comfortable for All-Day WearI can wear these all day without any discomfort. They’re light and fit snugly in my ears. A great fit for daily use."
- "I can hear much better with theseI can hear much better with these, but they sometimes feel a little loose after wearing them for a few hours. Other than that, I’m happy with the sound quality."
- "Finally I can listen to musicI love bluetooth feature, listening my favorite songs directly to my hearing aids feels amazing!!!"
- Robust features for a very affordable price
- Bluetooth streaming (iPhone and Android)
- Tinnitus masking features
- Requires some tech savvy
- Newer company
- Slightly less advanced sound (although impressive for the price)
- Elehear Beyond is an OTC product appropriate for mild-moderate hearing loss levels
- If you have more significant hearing loss you might consider local or Telehealth care. Read more about prescription Vs. OTC hearing aids here
- Available for purchase online or at retail stores
- Does not require a prescription from a doctor
- Customize your hearing aids through dials and controls inside a smartphone app
- Elehear Beyond has the best sound quality we've seen at products under $500
- Comfortable receiver-in-canal style is great for all day wear
- Sits behind the ear with a receiver (speaker) that reaches into the ear canal
- The form factor of Elehear Beyond is comparable to leading prescription hearing aids
- Only available in silver
- Elehear Beyond comes with rechargeable batteries and a portable recharge case
- 20 hours of wear on a single charge
- New recharge case carries an additional 80 hours of portable charge (without being plugged in)
- Elehear Beyond is compatible with iPhone and Android Devices
- Bluetooth connection is managed through the settings app
- Elehear Beyond is reasonably easy to handle but does require the wearer to place the hearing aid behind their ear and insert the receiver into the ear (consistent across all behind-the-ear styles)
- Elehear Beyond has an IPX5 rating
- In practical terms, an IPX5-rated device can handle splashes, rain, and some exposure to water but is not fully waterproof
- Adjust volume, sound quality and background noise
- Elehear Beyond comes with built-in tinnitus management features
Other OTC Hearing Aids Brands To Consider
Below are a few alternatives to the shortlist above. Alternatives include Apple’s AirPods Pro 3, Jabra Enhance Select, and Lexie B2 Plus. Each offers its own mix of features, price, and fit that may work better for certain users.
Jabra Enhance Select
- Bluetooth enabled and rechargeable
- 100-day risk-free trial
- Nearly invisible with a sleek design
- Professionally programmed before shipping to your home
The Jabra Enhance Select 700 offers an impressive blend of clinic-grade technology, affordability, and user convenience. Here’s why it stands out:
- Professional-Grade Technology at a Lower Price: Jabra’s Enhance Select 700 provides the advanced sound quality of clinic-only brands like ReSound but at less than half the price.
- Professional Programmed Hearing Aids From Home: Jabra’s pre-programmed approach ensures a personalized fit without needing in-clinic visits—customers simply take or upload a hearing test, and Jabra's audiologists program the hearing aids before shipment.
- Seamless Bluetooth Streaming: Equipped with LE Audio, the Enhance Select 700 delivers high-quality, direct streaming for calls and media, with excellent sound quality and range.
- Purchase Perks: Jabra offers 100-day returns, unlimited remote adjustments, and seven-day-a-week customer support.
Overall
Users commend the Jabra Enhance Select 700 for its natural sound quality, discreet design, and effective customer support. Many report significant improvements in hearing clarity and comfort.
Positives
- Natural Sound Quality: "The sound quality is excellent, and the ability to adjust settings via the app is a great feature." — Timothy T.
- Discreet Design: "These hearing aids are virtually invisible and very comfortable to wear throughout the day." — Angel M.
- Customer Support: "The customer service team was incredibly helpful and guided me through the setup process with ease." — Damian.
Complaints
While the majority of feedback is positive, some customers have reported issues with device durability and the need for adjustments. However, these concerns are relatively infrequent compared to the overall satisfaction expressed by users.
For a comprehensive view of customer experiences, you can visit Jabra Enhance's review page.
- One of the most affordable and convenient ways to purchase online while maintaining professional care
- Comes with a 100 day free-return period
- Offers rechargeable batteries and Bluetooth streaming
- Remote care requires users to be somewhat tech savvy
- Jabra only offers styles that sit behind the ear
- Jabra Enhance Select is a fully customizable, prescription hearing aid appropriate for mild-severe hearing loss
- If you have severe hearing loss and struggle with feedback (squealing) you may need a custom earmold (not available through Jabra Telehealth)
- If you have profound hearing loss, you may be a better fit for a high-powered BTE style hearing aid (note available through Jabra Telehealth)
- Jabra is a remote care brand that programs and delivers your hearing aids through Telehealth
- Take or submit a hearing test and a Jabra professional will program your hearing aids and ship them to your door
- Three years of unlimited support is included with every purchase
- Jabra Enhance Select hearing aids come with the capability for remote adjustments. This feature means you don't need to ship your hearing aids back to get program updates
- Jabra Enhance Select is manufactured by GN Hearing (the makers of ReSound hearing aids)
- Jabra devices come with a strong set of background noise handling features and app controls
- Jabra Enhance Select comes in a small and comfortable receiver-in-canal style that is easy to wear all day
- Sits behind the ear with a receiver (speaker) that reaches into the ear canal
- The form factor of Jabra Enhance Select is comparable to ReSound hearing aids
- Choose between five colors including: Sparkling Silver, Champaign, Gold, Bronze and Warm Grey
- Jabra Enhance Select offers three levels of technology. Select 500 and Select 300 come with rechargeable batteries and as of May 2024, Select 50 also comes with rechargeable batteries
- 30 hours on a full charge
- Comes with a portable charger (does not need to be plugged in)
- All Jabra Enhance Select technology levels allow Bluetooth streaming, music listening, and phone calls.
- Jabra Enhance Select 500 uses Bluetooth LE Audio (Auracast ready) and is handsfree for iPhone (11 and newer) and compatible Android models
- Jabra Enhance Select 300 allows iPhone users to take calls hands-free, while Android users must keep their phones close to pick up outbound audio
- Jabra Enhance Select 300 uses Apple's MFI connection and Android's ASHA connection to stream content through Bluetooth
- Jabra Enhance Select is reasonably easy to handle but does require the wearer to place the hearing aid behind their ear and insert the receiver into the ear (consistent across all behind-the-ear styles)
- If dexterity is a concern we recommend Select 300, which is larger and easier to grasp than the microRIE (Select 500)
- The Jabra Enhance Select charge case uses induction charging and is fairly easy to handle
- All levels of Jabra Enhance Select have an IP rating of 68
- These hearing aids can withstand dust and water submersion in one meter of water for up to 30 minutes
- Customization: Treble/bass, volume, microphone directionality
- Jabra Enhance Select includes Telehealth capabilities through the Jabra app
Eargo 8
- Disruptive leader in hearing health, known for sleek, invisible design.
- Rechargeable with a patented ear tip that allows ears to breathe.
- Eargo 8 adapts to your environment automatically throughout the day.
- Budget models (Eargo SE and Eargo LINK) offer lower prices and alternate features.
Eargo has long set the standard for invisible-style design in the hearing aid industry. Their compact devices, along with an intuitive app and thoughtful packaging, make Eargo a standout choice for early adopters. Here’s why we picked it:
- Invisible Design: The Eargo 8 is one of the smallest rechargeable hearing aids on the market, rivaled only by the Sony CRE-C20.
- Reduced Occlusion: Eargo’s patented “floating” ear tip design sits comfortably in the ear without causing occlusion, a common issue for in-ear hearing aid users.
- User-Friendly App: The Eargo app is easy to navigate, and it allows audiologists to remotely adjust your devices as needed, though most adjustments can be managed directly in the app.
Eargo has received numerous customer reviews, reflecting a generally positive reception.
Overall
Customers frequently commend Eargo for its discreet design, sound quality, and responsive customer service. Many users highlight the significant improvement in their hearing experience and the comfort of the devices.
Positives
- Discreet Design: "The Eargo hearing aids are virtually invisible, and no one can tell I'm wearing them." — James D.
- Sound Quality: "The clarity of sound is remarkable; I can hear conversations clearly even in noisy environments." — Charlie B.
- Customer Service: "Eargo's support team was incredibly helpful and guided me through the setup process with ease." — Rowdy G.
Complaints
While the majority of feedback is positive, some customers have reported issues with device durability and the need for adjustments. However, these concerns are relatively infrequent compared to the overall satisfaction expressed by users.
For a comprehensive view of customer experiences, you can visit Eargo's review page.
- Eargo is a tiny, rechargeable device that sits entirely inside the ear
- Eargo uses a unique design that prevents occlusion
- Eargo is self-fit using an app but offers remote support
- Only appropriate for those with mild to moderate hearing loss
- Requires some tinkering to fine tune the product
- Eargo is not Bluetooth enabled due to the tiny size
- Eargo 8 hearing aids are sold over the counter and are appropriate for those with mild-moderate hearing loss
- If you have more significant hearing loss it is recommended that you access professional care through Telehealth or local care
- Available for purchase online or at retail stores like Verizon Victra and Best Buy
- Does not require a prescription from a doctor
- Customize your hearing aids with an onboard hearing test and app-controls
- Eargo has well-developed background noise management algorithms
- Smart Sound Adjust automatically adapts to your environment and focuses on voices
- Eargo sits inside of the ear and is one of the most comfortable in-ear options on the market
- Eargo creates less occlusion than many in-the-ear options
- Our team finds all in-the-ear models slightly less comfortable than RIC hearing aids
- Sit inside the ear
- Only available in one completely-in-canal size (nearly invisible in many ears)
- Only available in black
- Eargo hearing aids are rechargeable
- 16 hours of battery life on a single charge
- Each pair of hearing aids comes with a small and portable recharge case that carries 14 additional charges
- Eargo 8 does not offer Bluetooth streaming
- Eargo 8 is small but comes with rechargeable batteries which prevents difficult battery changes
- Eargo's devices include a pull-tab that makes it easier to place and remove the devices in your ears
- Eargo 8 hearing aids have an IP68 rating, meaning they can be submerged in up to one meter of water for 30 minutes without damage.
- Eargo 8 pairs with a smartphone app that includes an onboard hearing test and manual controls for programs, volume, and sound quality.
- You can also adjust volume and switch programs without the app by simply double-tapping your ear.
Lexie B2+ Powered By Bose
- Self-fit with the Lexie app, adjusting treble, bass, and directionality.
- Rechargeable with 18-hour wear time and an on-the-go charging case.
- Bluetooth streaming for calls on both iOS and Android.
- On-demand support from Lexie hearing experts for setup and care.
Lexie leads the OTC hearing market in volume, with wide availability in pharmacies and strategic partnerships that make it highly accessible. Here’s why it stands out:
- User-Friendly App with Extensive Adjustments: Lexie’s app offers near-limitless adjustment options, ideal for users who want full control over their sound quality.
- Top-Notch Customer Service: Lexie excels in customer support, allowing users to connect quickly through the app for any assistance needed.
- Soundly Extras: Purchasing Lexie hearing aids through Soundly includes a complimentary audiology setup call, ensuring you start with a perfectly tailored fit.
Ease of Use and Setup
- Cindy B. – "After a lot of research on OTC hearing aids for my moderate hearing loss, I chose Lexie B2 Plus. I wanted rechargeable hearing aids that would link to my iPhone, and the price was affordable...One of the most exciting experiences I’ve had was hearing the birds in my backyard! It was almost completely quiet without them on."
- William – "These Lexie B-2's were easy to adjust per the app and proved to be superior to the more expensive ones, plus no trips to the doctor every week or so."
Sound Quality and Listening Experience
- Faith Li – "The Lexie B2 hearing aid doesn't get time to charge as I just love wearing them. I am grateful for the gift of hearing and that I no longer miss out on the moments."
- Listener T. – "The sound quality through the aids was fine, once I had them set appropriately. The noisy room filter allowed me to focus on the person in front of me in a restaurant."
Affordability and Value
- Carolyn – "I kept putting off getting hearing aids because they are so expensive and my insurance doesn’t cover them. Once the audiologist mentioned the high quality, I looked into them...What a difference they have made!"
- Kyle, MD – "The Lexie B2 Plus hearing aids are very good introductory hearing aids...The sound quality is excellent and extremely natural."
Challenges
- DJR-NJ – "Customer service and support were excellent! Unfortunately, I was unable to wear the devices because the wires were too short. They did respond quickly to my calls and sent the longest wires they make, but I still couldn't wear them long enough to judge their performance."
- James - "Android App on my phone kept dropping connection with the Lexie hearing aids. The app was more consistent on my iPad"
- Shopper A - "Android phone tries to play audio through hearing aids with No Way To Turn it off unless it's a phone call. Otherwise, you must turn your media volume off. Phone call through hearing aids mutes your microphone so caller can't hear you."
- Strong underlying technology
- Self-fitting using the Lexie app
- Affordable and rechargeable
- Only comes in one color
- Not for those with severe hearing loss
- Lexie B2 Plus - Powered by Bose is an OTC product appropriate for mild-moderate hearing loss levels
- If you have more significant hearing loss you might consider local or Telehealth care. Read more about prescription Vs. OTC hearing aids here
- Available for purchase online or at retail stores like Walgreens and Best Buy
- Does not require a prescription from a doctor
- Customize your hearing aids with an onboard hearing test and app-controls
- Lexie B2 Plus Powered by Bose is among our top-rated OTC hearing aids for overall value
- Technology from Bose does a great job managing background noise
- Comfortable receiver-in-canal style is great for all day wear
- Sits behind the ear with a receiver (speaker) that reaches into the ear canal
- The form factor of Lexie B2 Plus Powered By Bose is comparable to leading prescription hearing aids
- Only available in grey
- Lexie B2 Plus Powered by Bose comes with rechargeable batteries and a portable recharge case
- 18 hours of wear on a single charge
- New recharge case carries an additional 18 hours of portable charge (without being plugged in)
- Lexie B2 Plus Powered by Bose added Bluetooth streaming for iPhones in 2023 and now with compatible Android™ devices
- Refer to the FAQs at LexieHearing.com to check if your Android™ device is compatible.
- Lexie B2 Plus Powered by Bose uses Apple's MFi protocol for connection
- Streaming functionality is designed for phone calls. While music and media streaming is possible, it is not optimal.
- Lexie B2 Plus Powered by Bose is reasonably easy to handle but does require the wearer to place the hearing aid behind their ear and insert the receiver into the ear (consistent across all behind-the-ear styles)
- Lexie B2 Plus Less waterproof than industry leaders but still sweat and moisture resistant
- Take an onboard hearing test to customize your hearing aids
- Adjust directionality between “Everywhere” and “Front” modes so you can choose between hearing sounds around you or the sound that’s in front of you
- Manually customize treble/bass, volume and programs
Over-The-Counter Hearing Aid Comparison Table
Over-The-Counter Hearing Aid History
The concept of buying hearing aids directly, without a doctor’s visit, is relatively new. The over-the-counter (OTC) hearing aid category was officially created in late 2022 by the FDA after years of bipartisan advocacy and collaboration across multiple presidential administrations.
The goal was simple: increase hearing aid adoption. Wearing hearing aids is one of the most effective ways to improve overall health, reducing risks associated with falls, social isolation, earnings loss, and even dementia. Hearing aids are safe and highly effective for most users.
- The push began in 2015, when the President’s Council of Advisors on Science and Technology (PCAST) sent a letter to President Obama urging action.
- In 2017, Congress passed the Over-the-Counter Hearing Aid Act of 2017, laying the foundation for the category.
- From there, the FDA spent years designing a framework that would make sense to consumers while fostering competition in the hearing aid industry.
- Finally, in 2022, the FDA published its guidelines. The 200-page document capped the maximum volume output for OTC hearing aids to ensure consumer safety making OTC hearing aids best for those with mild-moderate hearing loss.
New Brands Quickly Entered The Category
The finalized guidelines sparked a wave of new brands and products, which continues to evolve. Interestingly, by 2026, the largest prescription hearing aid manufacturers also lead the OTC market—such as Sonova with Sennheiser and WSA with Sony.
For those curious, we’ve included a full history in the FAQ section of this guide.
Are Over-the-Counter Hearing Aids Right for You?
At Soundly, we’ve helped thousands of people navigate their hearing aid options, and one of the most common questions we hear is: Are over-the-counter (OTC) hearing aids right for me?
If you’ve already had your hearing tested, that’s a great first step. If not, we encourage you to take Soundly’s hearing test. It only takes about five minutes, requires a pair of headphones, and will give you a clear picture of the severity and shape of your hearing loss.
If you’d like a second opinion after taking the test, reach out to our team at 1-833-SOUNDLY or email us at [email protected]. Watch the video below for a full review on how to read your test.
Prefer to watch? Here's an in depth tutorial on how to read your hearing test. 👇
How to Decide Between Rx and OTC
Here are a few key considerations to keep in mind when choosing between hearing aids purchased at a local clinic and those purchased over the counter.
- Severity of Hearing Loss
OTC hearing aids are designed for mild to moderate hearing loss. The FDA caps the maximum volume output at 117 dB SPL, which is plenty for many people but might not be enough if you have severe or profound hearing loss. In those cases, a prescription device fitted by a professional may be a better option. - Appetite for Self-Fitting
OTC hearing aids require some level of tech comfort. Leading brands like Sony and Sennheiser are designed to be easy to set up, and when purchased through Soundly, you’ll receive a complimentary audiologist setup call. Still, you’ll need to download a smartphone app, make occasional adjustments, and ensure the device is set up correctly. If you prefer face-to-face support in a clinic, prescription hearing aids may be worth the extra cost. - Insurance Coverage
Insurance coverage for hearing aids is limited and even rarer for OTC devices. Most insurance policies that cover hearing aids offer discounts on prescription models rather than coverage for OTC options. This means OTC hearing aids are usually a cash purchase - you can use your HSA and FSA dollars. While more affordable than prescription devices, it’s worth planning for this upfront.
Comparing OTC and Prescription Technology
Many OTC products don’t meet our standards, but several of the brands on this list deliver quality comparable to what you’d find in a clinic. In fact, some are even made by the same parent manufacturers, often on the same production lines (see Sony and Sennheiser).
Comparing OTC and Prescription Setup
The key difference lies in how your devices are programmed: prescription devices are programmed by a professional, while OTC devices rely on your adjustments using an app.
Ultimately, the decision comes down to your priorities. Do you value the convenience and lower price of OTC hearing aids, or is support in a doctors office worth the higher cost of prescription devices?
Taking our hearing test is a great first step in deciding what’s right for you. We’re here to help you make a confident choice.
How to Choose an OTC Hearing Aid

If you’re new to the OTC hearing aid market and have determined that you’re a good candidate, you might be wondering how to choose from the dozens of available options—or even just the few listed in this guide.
We recommend focusing on three key factors: style, features, and support.
1. Style
OTC hearing aids generally come in three styles: behind-the-ear, in-the-ear or open-ear.
- Behind-the-Ear
Behind-the-ear hearing aids are the most widely adopted because they’re discreet, comfortable for all-day wear, and typically come with advanced features like Bluetooth streaming and additional microphones. Since the main device sits behind your ear, the piece inside your ear canal is smaller, adding to the comfort. If streaming and sound quality are your priority, take a look at Sennheiser All Day Clear. - In-the-Ear
Some people prefer in-the-ear hearing aids because they’re out of the way for mask-wearing, hearing protection, or simply because they feel more discreet. In-ear devices like the Eargo 8 offer excellent sound quality and are becoming increasingly compelling options. However, one trade-off with invisible styles is that they typically don’t support Bluetooth streaming. - Open Ear (New in 2025)
Until 2025, there were no true hearing aid options that didn’t require something in your ear. That changed with the launch of Nuance Audio, which uses directional microphones to capture sound from the direction you're facing and deliver it back into your ears.
2. Features
Once you’ve decided on a style, consider which features matter most to you:
- Rechargeable Batteries
Rechargeable batteries have become standard in the hearing aid industry. All the products in this guide come with rechargeable batteries and at least 16 hours of battery life per charge, ensuring they last a full day before needing a recharge at night. - Bluetooth Streaming
If you’d like to use your hearing aids as headphones for streaming music or taking calls, focus on behind-the-ear models with Bluetooth capabilities. Invisible-style hearing aids don’t yet support Bluetooth streaming. - Smartphone Apps
Another important feature to consider is the smartphone app that comes with your hearing aids. The best apps allow you to test your hearing, adjust frequencies and volume, and fit to your hearing loss. Each of the products in this guide feature simple and robust apps, but each has its own style. Be sure to check our in-depth reviews to find an app that suits your needs and comfort level.
3. Support
Finally, think about how much support you’ll need during setup and use. If you purchase through Soundly, you’ll receive a complimentary setup call with our audiologist and ongoing support from our team.
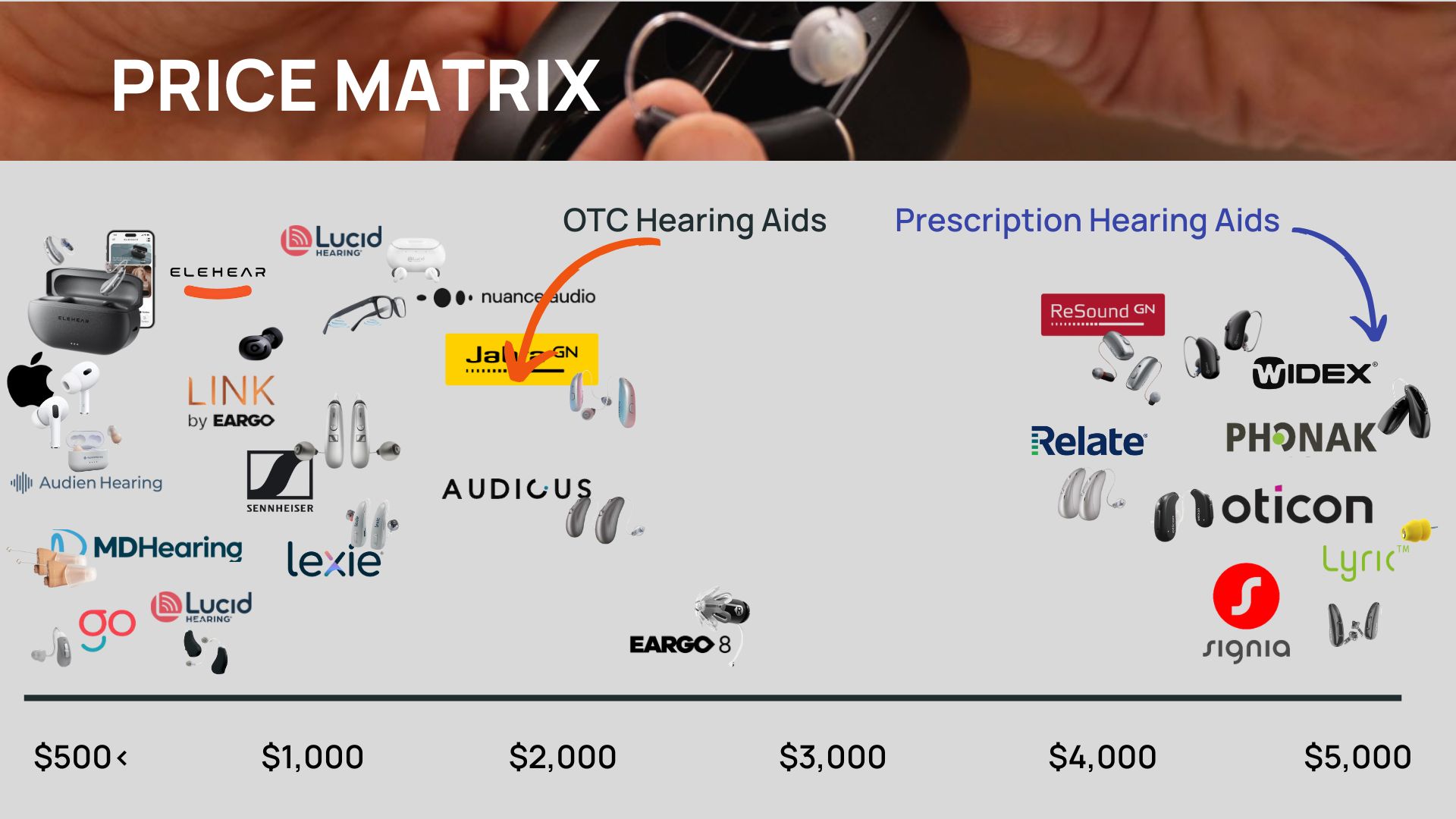
OTC Hearing Aid Prices: What to Expect
OTC hearing aid prices range from about $100 to $2,000, making them significantly more affordable than prescription hearing aids, which average $4,500 per pair. But what drives the price differences within the OTC category? Broadly, we see three pricing tiers:
- Budget-Friendly Options ($100–$500)
Most hearing aids in this range do not meet our quality standards. We’ve tested dozens, and many are uncomfortable to wear or simply ineffective. Reviews on platforms like Amazon and Best Buy echo these concerns. However, there are exceptions. After extensive testing, we’ve found the Elehear Beyond at $399 to be a reliable and affordable option. - Mid-Tier Options ($800–$1,200)
This price range offers the best value for most people. Products like Sennheiser, and Nuance Audio deliver high-quality performance at a fraction of the cost of prescription models. These devices are backed by trusted manufacturers and feature technology comparable to what you will find in a clinic. For many, this is the sweet spot for balancing quality and affordability. - Premium Options ($1,500–$2,000)
At the higher end, brands like Jabra Enhance Select and Eargo justify their price with added features. Jabra, for instance, includes professional programming before your devices are shipped, while Eargo invests heavily in R&D to create nearly invisible hearing aids. These products cater to those seeking extra convenience or cutting-edge design.
While prices vary widely, the $1,000 range is where most people find the right balance of cost and quality.
Final Thoughts

If you’ve made it this far, take heart—you now have the information you need to determine if you’re a good candidate for OTC hearing aids and how to choose the right product.
After testing more than 50 hearing aid brands, I’m genuinely encouraged by the progress in the OTC market over the past two years. Some of today’s leading OTC brands are comparable to top-tier prescription options—a fact confirmed by studies published in the Journal of the American Medical Association (JAMA).
OTC hearing aids offer clear advantages: they save you time by skipping the need for a doctor’s visit and can significantly reduce costs. That said, they’re not the right choice for everyone. If you have more severe hearing loss or prefer the in-person guidance of a local audiologist, a prescription option may be a better fit.
Beyond my own testing, I’ve had the privilege of seeing our team at Soundly support thousands of users in their journey to better hearing with OTC devices.
Time and again, we hear the same thing: those who address their hearing loss feel more confident and engaged with the world around them.
To us, that’s a win—and we hope it will be for you too.
Frequently asked questions
October 2015 - PCAST Letter Sent to President Barack Obama
OTC hearing aids got a significant boost from the President’s Council of Advisors on Science and Technology (PCAST) in 2015. PCAST recognized that hearing loss was a recurring issue prominently brought up by older adults. They noted that the traditional medical requirement to obtain hearing aids was a barrier for many individuals who needed them. In response, PCAST advocated for the removal of this medical prerequisite. Their efforts began the process to the creation of a new category of OTC devices, designed to be easily accessible and affordable for those with mild to moderate hearing loss.
December 2016 - FDA Takes The First Step
Thirty-nine years after the FDA’s first ruling on hearing aids, the agency came back with a blockbuster announcement in December 2016 that it would no longer enforce the requirement that individuals 18 and up receive a medical evaluation or sign a waiver before purchasing most hearing aids.
In that same announcement, the FDA shared their “commitment to consider creating a category of over-the-counter (OTC) hearing aids that could deliver new, innovative and lower-cost products to millions of consumers.”
March 2017 - Elizabeth Warren and Chuck Grassley Introduce a Bill
In May 2017, Democratic Senator Elizabeth Warren and Republican Senator Chuck Grassley signaled a bipartisan commitment when they introduced the Over-The-Counter Hearing Aid Act of 2017. In May, the two lawmakers co-authored a piece that was published on the Jama Network. They urged the FDA to “ go further than simply doing away with the medical evaluation requirement and create a new category of over-the-counter hearing aids for adults with mild to moderate hearing loss.”
July and August 2017 - Congress Says Yes
On July 12th, 2017, the bill earned bipartisan support in the U.S. House. Three weeks later, on August 3rd, the Senate passed the bill to the president’s desk.
August 18th - President Donald Trump Signs The Bill
On August 18th, 2017, President Trump signed the bill into law. The law mandated that the FDA create and regulate a new category of OTC hearing aids. The bill directed the FDA to focus on high standards for safety, consumer labeling, and manufacturing.
2017 through 2021 - The Waiting Game
After a lot of initial debate and speculation, everything went quiet. The FDA went to work on its regulations, and hearing aid wearers had no choice but to wait.
The three-year deadline loomed at the beginning of 2020, and it looked like we would finally get answers. Then came the pandemic. The FDA had bigger things to worry about, and OTC hearing aids got stuck on the back burner.
July 2021 - President Joe Biden Signs An Executive order
After nearly four years of crickets, President Biden lit a fire under the issue On July 9th with an executive order that directed the U.S. Department of Health and Human Services “to consider issuing proposed rules within 120 days for allowing hearing aids to be sold over the counter.”
October 2021 - At Long Last, A Proposal
On October 19th, the FDA released the long-awaited proposal for a new class of over-the-counter hearing aids. The 114-page document was long and technical (as it should be). As a part of the brand new guidelines, the FDA established a 90-day comment period where consumers, companies, and doctors could submit their feedback on the bill.
January 2022 - Comment Period Closes
In late January, the comment period closed, with more than 1,000 comments submitted from audiologists, manufacturers, and even hearing aid wearers. As you might expect, the disruptive regulation has been contentious, with many advocates and detractors on both sides.
August 16, 2022 - Final Guidelines Are Published
After reviewing more than 1,000 comments from manufacturers and consumers, new guidelines finally published. The final FDA ruling is over 130 pages long and includes in-depth responses to comments on topics like labeling, applicable technology, the OTC application process, and more. The final ruling made several changes in response to industry comments but largely remained intact.
October 17, 2022 - OTC Officially Go OTC
Starting on October 17th consumers began to see officially labeled OTC hearing aids on shelves at retailers like Walgreens, Best Buy and online. OTC hearing aids can now be purchased without a doctors appointment and will allow fitting at home.
Someone buy Elizabeth Warren and Chuck Grassley a drink. 🍺
October 2015 - PCAST Letter Sent to President Barack Obama
OTC hearing aids got a significant boost from the President’s Council of Advisors on Science and Technology (PCAST) in 2015. PCAST recognized that hearing loss was a recurring issue prominently brought up by older adults. They noted that the traditional medical requirement to obtain hearing aids was a barrier for many individuals who needed them. In response, PCAST advocated for the removal of this medical prerequisite. Their efforts began the process to the creation of a new category of OTC devices, designed to be easily accessible and affordable for those with mild to moderate hearing loss.












